

Research at RMES seeks to improve energy storage and conversion performances using nanoscience as a leverage to drive specific reactivities and enhance ion transport at the electrode/electrolyte interface. We are interested in various technologies such as all solid-state Li-ion, flow and metal-air batteries, proton exchange membrane- and solid oxide- fuel cells, and solar hydrogen production.
Electrodes, electrolyte or combo materials are prepared as nanostructured and hybrid materials, using sol-gel chemistry or electrochemistry, combined with the appropriate processing technique (dip-coating, spray-coating, electro-spinning). To improve their performance in the device, we investigate the processes at the interfaces using electrochemical methods coupled with ex-situ and in-situ X-ray-based and electron-based spectroscopy.
Highlights:
Photoelectrochemical Energy
One of the major roadblocks to large-scale usage of solar power is the storage of energy during periods of little to no sunlight. One possible solution is the direct conversion of sunlight into chemical fuels (hydrogen) or into ion storage. Photogenerated charge carriers can be used to promote a redox reaction, thus storing solar energy in the form of chemical bonds or ions stored into a crystallographic structure. In order to promote this conversion, the electron-hole pair must be separated – typically by an electric field within the device. This places certain requirements on photoelectrode materials; namely, that they exhibit high charge carrier mobility and good light absorption in the solar spectrum.
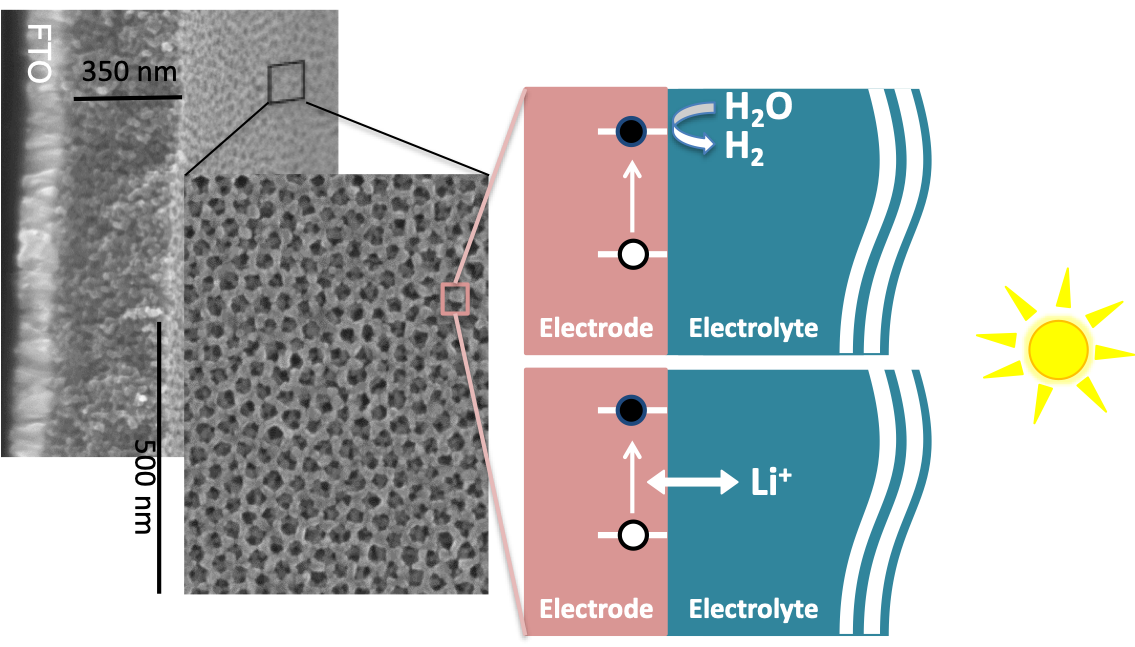
Schematic of Illuminated semiconductor-electrolyte junction for water splitting and photobattery
Hybrid Materials for Clean Energy
The application landscape for electrochemical energy storage technologies is set to expand rapidly over the next several decades as demand grows in new areas ranging from micro-devices to electrified transportation and clean energy storage and supply. Several unique characteristics of hybrid materials make them ideal candidates for combining high energy and power at the material level. These advantages include: (i) increased electrochemically active surface areas for charge transfer, (ii) reduction of ionic transport resistance at smaller diffusion length scales, and (iii) the ability to incorporate ionic materials into a nanostructured framework capable of sustaining high powers.
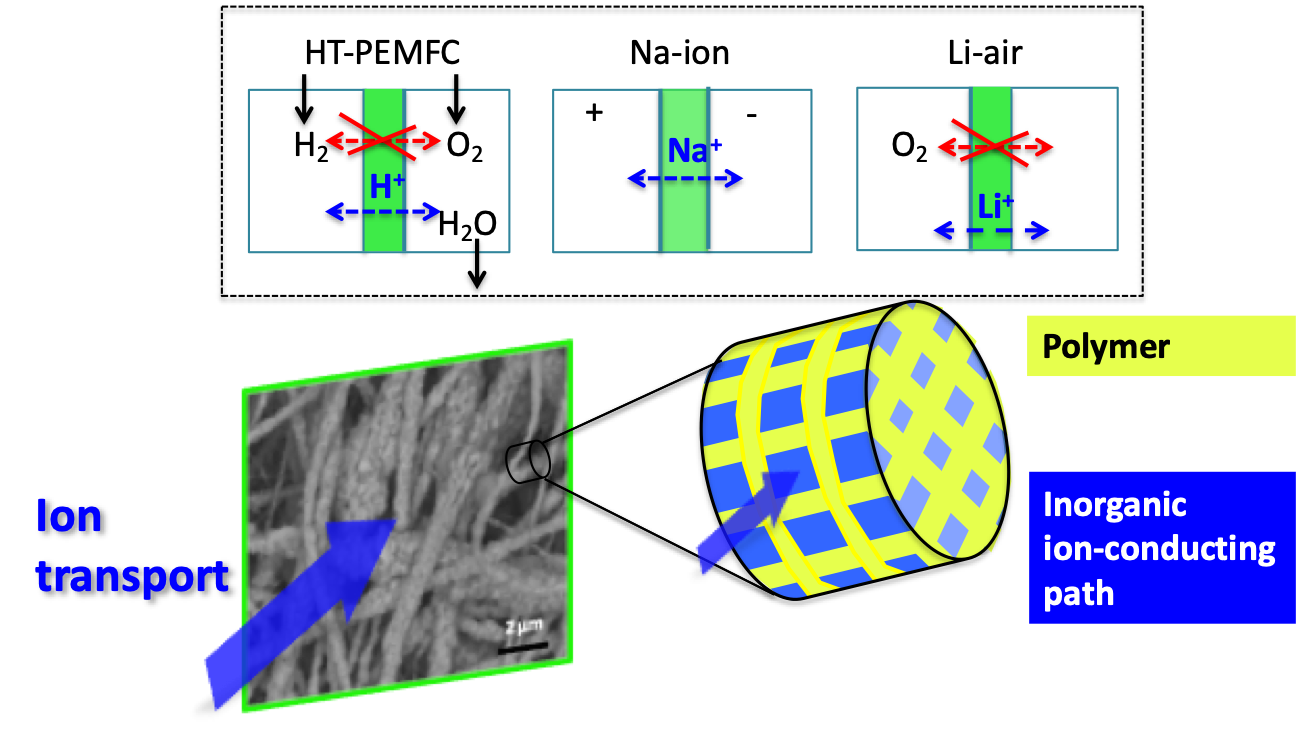
Schematic of Hybrid Organic-Inorganic Membranes for i) Proton Exchange Membrane Fuel Cell and ii) Separator for Na-ion Battery, iii) Membrane for aqueous Li-air battery
Interests:
1. Electrochemical Energy Storage and Conversion
2. Catalysis/Electrocatalysis/Photoelectrocatalysis
3. Nanoscience
Collaborations:
Internal
Electrocatalysis : design of Nanomaterials through soft chemistry approach
David Portehault, Olivier Durupthy, Sophie Carenco, NanoTeam
Film Processing and Characterization by Ellipsometry (ellipsometry porosimetry and thermoellipsometry)
Cédric Boissières, Marco Faustini, MHP team
External :
The LCMCP belongs to the French Research network on electrochemical energy storage, RS2E
Characterization of Multiscale Hybrid Organic-Inorganic Membrane at different Lenght Scale by using SANS, QENS, SAXS-WAXS techniques
Manuel Maréchal, Sandrine Lyonnard, Gérard Gébel, CEA-Grenoble
Mechanical Properties of Ultrathin Composite Membranes
Arnaud Antkowiak and Sébastien Neukirch, Institut Jean Le Rond d’Alembert, Sorbonne Université
Coupled fast quartz crystal microbalance (QCM) and electrochemical impedance spectroscopy to Study transfert at the electrode/electrolyte interface
Ozlem Sel and Hubert Perrot, LISE, Sorbonne Université
Solid State Chemistry, electrocatalysis, Na-ion and Li-air batteries
Alexis Grimaud, Gwenaëlle Rousse and Jean-Marie Tarascon, Collège de France
Collagen-based electrospun membranes
Carole Aimé, ENS
Grazing incidence XRD of mesoporous thin film on FTO substrates
Gianguido Baldinozzi, SPMS, CNRS CentraleSupélec, Université Paris-Saclay
